How to solve aging
What the longevity field is missing
There’s an odd gap in the longevity discourse:
Any coherent framework for how we will solve aging requires therapeutics simultaneously capable of both complex effects and permeating well through the entire human body.
Yet no such modalities currently exist. And this does not seem to be a major focus of aging research. Why?
Modern modalities
Current therapeutic modalities generally fall into one of two camps: systemically distributed but mechanistically limited, or mechanistically powerful but distribution limited.
Small molecules can access all tissues, organs, cells, and sub-cellular compartments, but are basically fairly crude tools1 that usually break things.2
Large “molecules”—CRISPR, gene and cell therapies, antibodies, biologics, etc—can effect much more complex transformations upon drug targets, but are very limited in terms of what parts of the human body they can actually access.3
No current modalities are both mechanistically powerful and systemically distributed.
An aging thesis
The thesis of this essay is two-fold:
i) we will not solve aging until we develop systemic, cell and CNS permeable, therapeutics capable of achieving complex transformations upon drug targets,
and
ii) Induced Proximity Medicines (IPMs) are the only credible shot of achieving this in the near future.
Here’s why.
The future of longevity is induced proximity
IPMs are a class of medicines that work through the action of bringing two macromolecules together, generally a target protein and an effector protein, to drive some therapeutic outcome (very different from how a typical therapeutic works). This can be called a protein-protein interaction or more simply an (induced) “proximity event”.
All biological processes are downstream of binary protein interaction events, so gaining specific control of proximity events theoretically enables a nearly unlimited set of biological outcomes.
While IPMs can be either small or large molecules, the most powerful examples are small molecules, as they retain their oral bioavailability and systemic distribution, and in this piece IPMs refers only to these.
IPMs are both a new and old class of medicines. Old in that the greatest chemist of all, Mother Nature, often makes molecules that work through induced proximity.4 Also old in that many human-developed drugs have serendipitously been found, many years later, to work through induced proximity.5 But new in that we are just learning to design these molecules ourselves rationally, and tap into the massive new pharmacology space they offer.
Why do they make so much sense for longevity?
There are two main processes we need to modulate systemically in humans for which induced proximity medicines are especially relevant: proteostasis and transcription.
Rescuing proteostasis
Proteostasis is the body’s ability to maintain a healthy, functional proteome, and dysregulated proteostasis is one of the most well-established hallmarks of aging: as we age misfolded proteins accumulate, the ubiquitin-proteasome system (UPS) loses efficiency, autophagy declines, and chaperone capacity becomes overwhelmed.
Proteostasis dysregulation isn’t a localized problem; it happens in every tissue, cell, and organ in our bodies, and it is causative or implicated in many aging-associated pathologies: neurodegeneration, amyloidoses, tauopathies, sarcopenia, and immunosenescence, to name just a few.
How is proteostasis regulated? Via proximity events—protein interactions of proteostasis machinery with dysfunctional or misfolded proteins.
Restoring proteostasis at scale requires the ability to selectively recycle, refold, or redirect specific proteins across the entire human body.
IPMs that induce productive interactions between proteostasis machinery and dysregulated proteins, or that directly tune the machinery itself, are the best shot we have of achieving this.
Root access to human transcription
Most cells in the human body contain our entire genome, but in any given cell only a fraction of those genes are transcribed into RNA, then translated into proteins. This is the dynamic process by which our bodies grow, respond, and adapt to various stimuli.
The key players are transcription factors (TFs). TFs are proteins that bind to specific DNA sequences to induce transcription of target genes and genetic programs. Any given TF can bind to many sites of the genome, and the process by which this is determined is entirely regulated by protein interactions. A given TF will have hundreds or thousands of interacting partners capable of tuning its function in response to various stimuli.
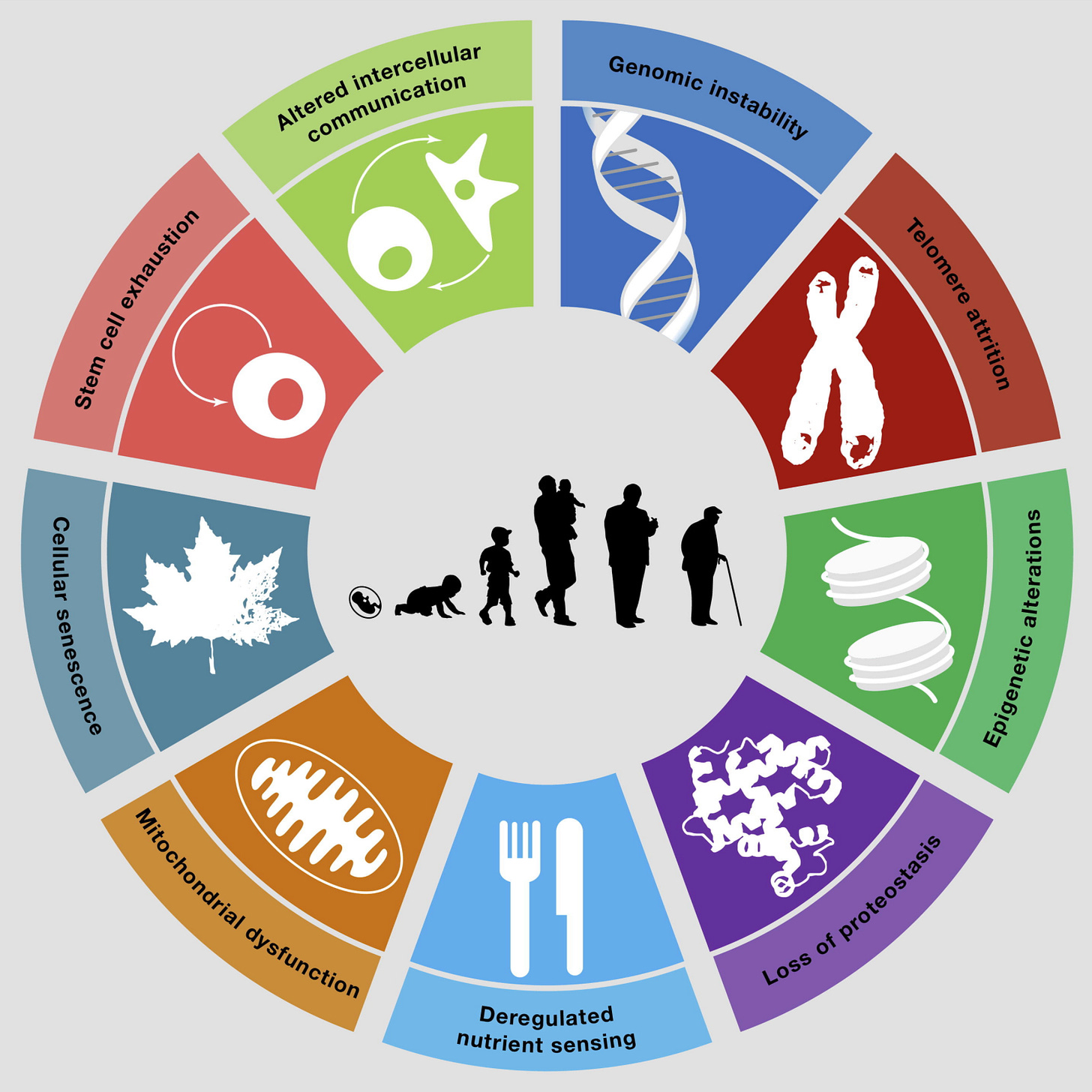
All of the hallmarks of aging, including proteostasis, are regulated via transcription.
TFs are a canonically “undruggable” class of proteins due to their often unstructured nature, and lack of traditional small molecule binding sites.
However, two factors make TFs much more amenable to modulation via IPMs:
physiologically they are controlled by protein interactions678, so if one can find the right proximity partner, one can gain far more control of TF biology than a normal therapeutic
even intrinsically disordered proteins like TFs become folded in certain contexts, most notably when in complex with another protein or macromolecule
By finding the right partner for a given TF, meaning one that both induces a productive and druggable complex, and then inducing that proximity event with a small molecule, we can develop systemic therapeutics capable of transcriptional modulation and reprogramming for the most important diseases.
We won’t solve aging until we achieve root access to human transcription.
A note on gene therapies for transcriptional reprogramming
To date, the primary way researchers have attempted to modulate transcription has been through gene therapies.
The majority of the cellular reprogramming research to restore youthful phenotypes or cellular functions relies on genetic delivery of TFs to aged cells.
Data generated in these studies have shown that, at least in vitro, it is possible to reprogram human cells to a more youthful state.
However, there are three major problems with using gene therapies to reprogram human cells in the clinic:
Delivery: we are very far away from being able to deliver genes to the majority of cells and organs in the human body.
Transcriptional control: very importantly, bulk delivery of a TF to a human cell does not phenocopy how either our bodies or IPMs normally regulate TFs. Physiologically, TFs are controlled by protein interactions (proximity events) which is quite different from just grossly increasing TF level. TF abundance ≠ TF activity.
Safety: gene therapies have a relatively long duration of action, which is unlikely to be ideal for safely and systemically reprogramming human cells. A short pulsatile bolus of transcriptional modulation, achievable by IPMs, is likely to be significantly safer, and is again much more similar to how transcription is regulated physiologically.
Even for liver-targeted gene therapies, where delivery is much less of an issue, I suspect it will be very difficult to get aging therapeutics approved in otherwise healthy humans. It’s an extremely important challenge, and luckily there are many smart people working on this.
Once and future IPMs
What do we need to do to get to this future? We are still in the dawn of the induced proximity era. Molecules like rapamycin are powerful proximity medicines, but very non-trivial to design or discover.
The first instance of rationally designed IPMs, PROteolysis TArgeting Chimeras (PROTACs), are nearing FDA approval, but are only capable of achieving a specific proximity event (E3-ligase mediated degradation) of a drug target, and not of more complex transformations.
To fully realize the promise of this field we need to build tools and technologies that let us understand exactly which proximity event we need to induce between the hundreds of millions of binary possibilities, and then allow us to develop small-molecule drugs recapitulating these proximity events.
My company, General Proximity, is hard at work on this, but to my knowledge there are no other longevity companies developing proximity medicines, which I find frankly baffling.9
Will IPMs actually “solve aging”? Of course we can’t know this, but with arbitrarily powerful control of transcription nearly anything is possible. I’d bet all my money that the way this will first become possible is through IPMs rather than some sort of next-gen gene therapy tech.
Hopefully this article will inspire many more researchers both in academia and industry to enter the IPM field, to help develop what I believe will be the single most important class of therapeutics in our lifetime.
The year is 2038
The year is 2038. Aevitum® hit the market five years ago after conditional Phase 2 approval for a rare mitochondrial disorder, but the biohackers were on it for years before now that the FDA’s right-to-try program only requires an AI doctor to perform a simple risk/reward analysis for each individual patient. Full approval for use in “healthy aging” was granted in 2036, making it the first ever rationally designed IPM to be approved for aging (and second ever aging IPM behind only rapamycin).
Aevitum® is also the first systemic transcriptional reprogramming medicine approved outside of cancer. It works by co-opting a key piece of intra-cellular machinery in order to activate a transcription factor involved in mitochondrial biogenesis—bringing these two biomolecules together in the exact right spatial orientation. The end result is that patients experience a significant increase in both total and healthy mitochondria, which drives a plethora of downstream benefits.
Analysts predict Aevitum® could be the first trillion dollar drug: roughly 0.5% of the world’s population now takes a once-monthly to once-quarterly pill at $500 USD a pop, via a self-serve DTC portal, and that number is expected to grow to nearly 4% in the next decade.
Elderly patients report energy levels they haven’t felt since the mid-20th century, with corresponding increases in mood and well-being (turns out having healthy mitochondria feels really good). In combination with 8th-gen GLPs, obesity is now entirely a matter of preference, rather than biology. Aevitum® is the drug of choice for Wall Street traders, and other knowledge workers, to increase their focus and energy, and the increased productivity it brings can be read out as a moderate uptick in the national GDP. Bryan Johnson’s nighttime erection scores have hit record highs.
Preliminary estimates are that Aevitum® will add three years of healthy lifespan to the average patient (four in America), while reducing risk for cardiovascular disease and various dementias by 30-45%.
New medicines for reprogramming other key transcription factors to support healthy aging are on the horizon, and the prediction markets have a number of long-term bets on who the first human to live past 150 will be.
While not (yet) a panacea, the affordable nature of such medicines has also begun to greatly decrease health inequities for tens of millions of humans.
The future is bright.
The entire history of drug discovery can be thought of as a reaction to this fact, where, in a series of paradigm shifting events, researchers developed more and more complex therapeutics enabling more complex transformations upon drug targets: antibodies, gene therapies, CRISPR, cell therapies, biologics, etc.
It’s a general physical principle that it’s easier to break something than it is to fix it. A normal small molecule is around 1% the size of its typical protein target (~500 Da vs ~50 kDa). Trying to use such a small molecule to fix or modify a target protein is possible (eg Vertex’s Trikafta), but is like asking an ant to fix a school bus engine, and in practice rarely works.
There is a huge amount of R&D focused on expanding the distribution limits of things like gene and protein therapies, but currently the ability to deliver such therapeutics to all cells and tissues in a human is sci-fi and seems likely to remain so for the foreseeable future.
EG auxin, rapamycin, and cyclosporin.
Thalidomide being by far the most famous example here.
An (incomplete) post-translational modification (PTM) map of p65 (part of NF-κB). There are ~200 types of PTMs possible in humans.

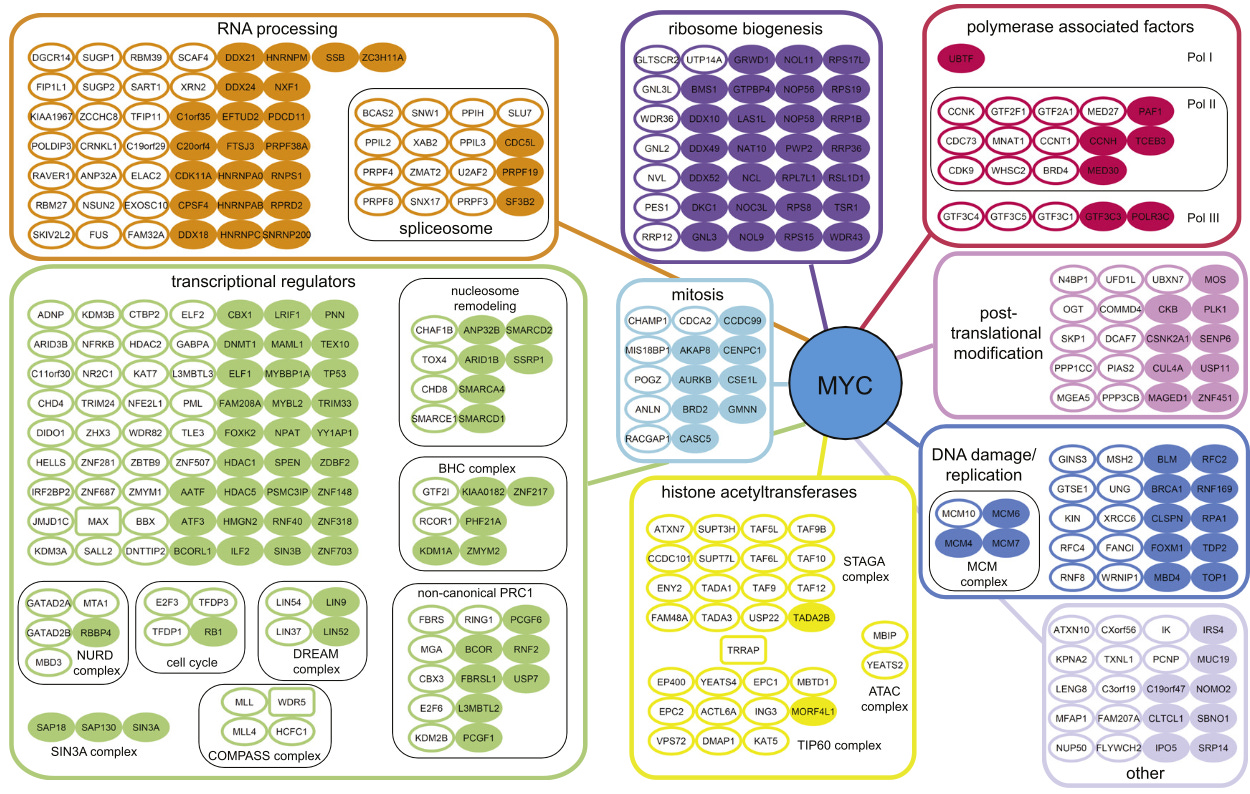
An (incomplete) interactome map for c-Myc.
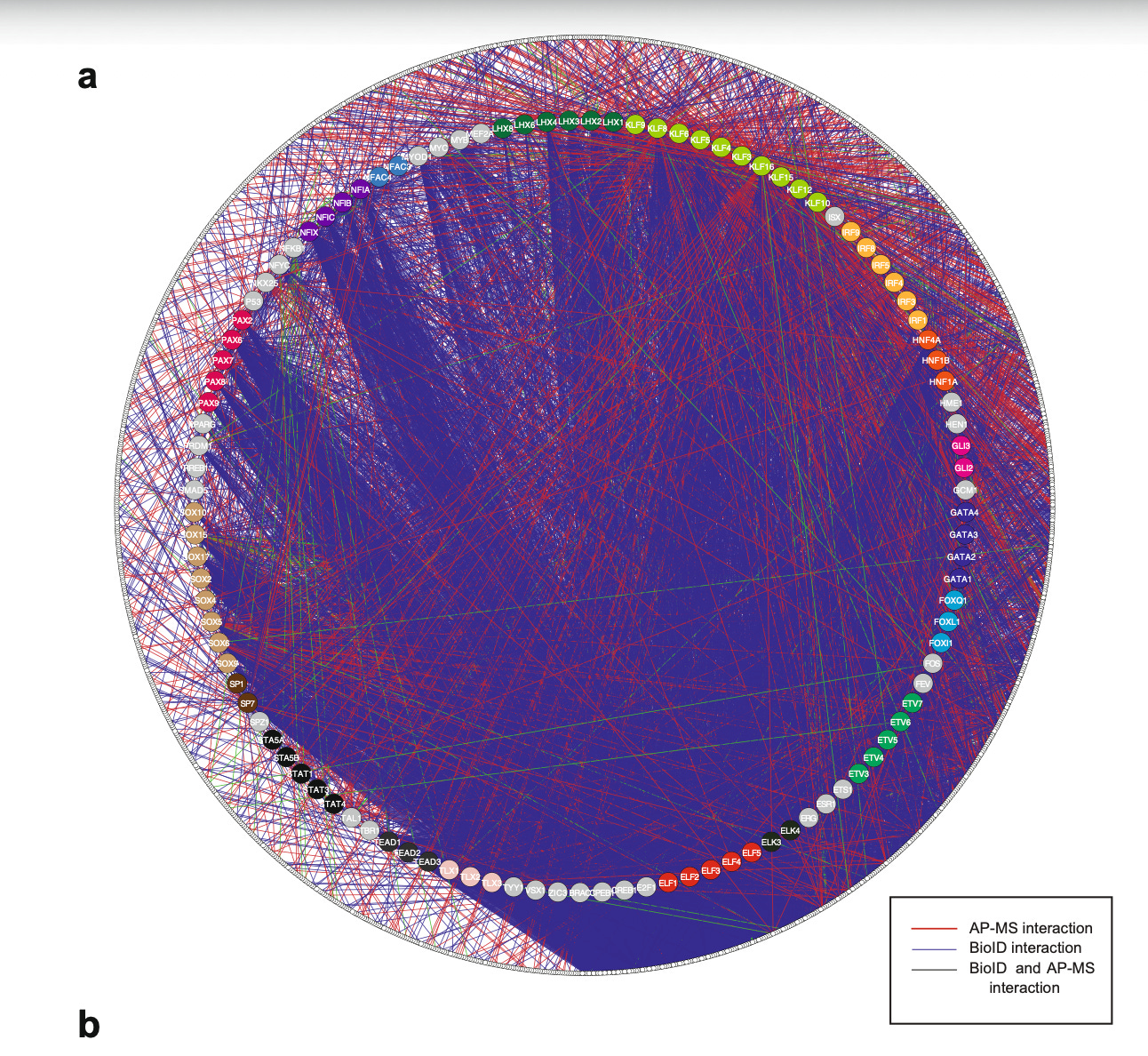
Human TF interactomes.
I’m sure there must be some out there, please say hi if that’s you. Also there are companies making new rapamycin analogues or developing degraders for things like tau that could be considered longevity proximity companies.

